Gas Fuels
Description
This section is from the book "The American Garden Vol. XI", by L. H. Bailey. Also available from Amazon: American Horticultural Society A to Z Encyclopedia of Garden Plants.
Gas Fuels
EVEN with the rapid spread of gas as a fuel for both heating and cooking, has come a very remarkable increase in the demand for gas. From letters received by The American Garden it is evident that great interest is taken by florists and owners of greenhouses in the matter of gaseous fuels. A number of florists already use natural gas in their greenhouses, and nearly all the manufacturers of steam and hot-water apparatus now furnish boilers adapted to burning gas. (See January American Garden).
It may be well, therefore, to ascertain, as clearly as we can without going into the technical side of the matter, just what is meant by gaseous fuel, and how such fuels should be used. There is, at first sight, an apparent confusion in the names of such fuels. There is natural gas, producer gas, fuel gas, illuminating and water gas, and there are a number of vapor fuels. These last are not gas fuels, but inflammable sprays or vapors made from oils. Some of these vapor and gas fuels are in greater or less degree of value in heating horticultural buildings.
We may begin with the oldest of the gases - common street or illuminating gas. The making of this gas is very simple and is at the same time expensive. For a long time it was the only method of making gas, and is still largely used in many of our cities. A coal, rich in gas, is placed in a retort over a strong fire and cooked. It amounts to cooking, but is properly called destructive distillation. That is, the gas is distilled out of the coal and the process destroys the coal, turning it into coke. The gas is cleaned or washed to free it from impurities, and is then stored in the holder ready for distribution through the street mains. This gas gives a luminous flame, and is a good fuel gas for warming greenhouses. The chief objection to it is the cost.
Natural gas is practically a wild gas found in the earth in vast quantities, and obtained by boring into the ground. It is an admirable fuel, and is used as such over a large extent of our country. Next to these come producer gas, water gas and fuel gas. These are technical terms, used to describe varieties of gas made in a number of ways in a great variety of gas-making plants that are commonly included under the general name of "gas producers." We need not here stop to examine these gases in detail. It is sufficient to know that coal, oil and. steam can be by some of these methods made to give us gaseous fuels that are suited to boilers for steam and hot water. The term water gas is misleading. Gas is not made directly from water. Steam that comes from water may be turned into a gas, but it is no longer steam, for it is completely decomposed and destroyed. Coal will also give gas, oils will also give gas, and these, combined with the gas made from steam, form, in various combinations, the various gases known as producer gas, water gas and fuel gas.
Our supply of coal and of oil for making these gases is apparently inexhaustible, and of water for steam, there is clearly no end. Many people have thought that if water can be turned into steam and then be destroyed and turned into a gas, that the supply of water might some day give out. Every gas lamp or gas stove that burns gives back to the atmosphere water. Nothing is really lost in the world. The elements change, but the amount of material in one form or another is forever unchanged. Fuel and water are undoubtedly the fuels of the future. They give good beat, and they are cheap. It is estimated that some of these gases can be made for less than 20 cents a thousand feet. We need never expect to buy them at this rate of the gas companies! Gas companies must have their fat little dividends, and it is doubtful if gas fuel will be sold at retail for less than 50 cents, including the delivery through the street mains. It is quite possible, however, that the time will come when florists with very large houses may set up gas producers on their own land, and make their own fuel, and in that case they will undoubtedly obtain very cheap fuel, as they will get rid of the costly mains laid in the streets.
Closely allied to these gas producers are the new apparatus for burning the spray or vapor of oil. These appliances, by means of a blast of steam or of compressed air or both, produce a fine spray or mist of petroleum oil, and this vapor may be burned under a boiler and give a very great heat. Some of these vapor producers are closely allied to gas producers, and their products seem in some instances to be a mixture of gas and vapor. Gasoline vapor stoves for cooking are also in use in a limited way, but it is doubtful if they are just the thing for heating greenhouses. Some of these vapor fuels are largely used in manufacturing and in steam making, and under various names may some day be of use in our greenhouses for steam boilers.
It is yet too early to say what is to be the outcome of these various processes, but the interest in the subject is so great, the demand for improved fuels so extensive, that out of them all is likely to come something that will be just what we want. So far as gas is concerned, it is not so much the fuel of the future as of to-day. Gas can be made in vast quantities at little cost. Almost every month sees new improvements in the gas-making plants and in new methods of producing gas. A non-luminous heating gas is the florists fuel - clean, safe, powerful, uniform and reliable. Only the cost stands in the way of its immediate universal adoption in our plant-houses. Every year, almost every month, sees something done to lower the price, and when the price falls gas fuel will come, and the long nights spent by the florist " tending fires " will be over forever.
How shall we burn gas ? Burners and "mixers,"as they are called when used with natural gas, are already in the market, and can be purchased very cheaply, or anyone can make one himself. The chief points are to give plenty of air for combustion and a good draft. In the Bunsen burner the air is mixed with the gas before burning, and this gives a larger volume of flame and a greater mass of hot air to flow through the pipes of the boiler. So far nearly all our gas stoves and gas heaters use the air and gas at the natural temperatures at which they may happen to be in at the time they burn. This is clearly not the most economical method. Both the gas and the air needed for combustion should be heated before they reach the burner.
The little drawing is designed to explain this matter of heating the air and gas before burning. The cylinder is closed at the bottom and opens at the top. The gas enters at the pipe marked 1, and passes downward to the burner, near the bottom of the cylinder. The air enters by the pipe 4, and passing downward escapes into the cylinder directly under the burner. At the side of the cylinder is a door for lighting the gas, and when the burner is started the door is closed. Now, both these pipes are over the gas flame and are heated by it. Both the gas and air are therefore hot when they reach the flame. The result is a longer, brighter and hotter flame; in other words, economy of fuel by causing the flame to give more heat for a given quantity of gas.
When any subject attracts general attention, and when many improvements are being made at the same time in any particular field of science, it always happens that new improvements are announced that promise to do something for nothing. Curious stories of wonderful gas-making machines, that will manufacture water gas in a cook-stove for nothing at all a thousand feet, have appeared in print. We can abide in peace, certain that while cheap gas is coming, it will be and must be made by scientific processes in regular plants erected for the purpose and on a large scale. The larger the plant the cheaper will the product be, and as florists and gardeners we can only wait till the gas manufacturers are ready to give us what we want - cheap fuel gas.
Charles Barnard.
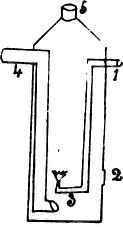
Diagram of Section of Ideal Regenerative gas-burner.
1. gas-pipe; 2, door for lighting; 3.burner; 4,air-pipe; 5, chimney.
Continue to:


