Of The Mechanical Division Of Bodies. Part 7
Description
This section is from the book "The London Dispensatory", by Anthony Todd Thomson. Also available from Amazon: PDR: Physicians Desk Reference.
Of The Mechanical Division Of Bodies. Part 7
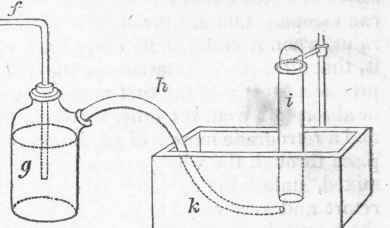
It is not required to be so large as the trough for water, and one part only need be sunk; the shelf should be on each side of this part, which is called the well; and it is useful to have an iron or brass stem, supporting a semicircular clip, fastened into the substance of the trough, to support the jar when it is filled with mercury and inverted. By this apparatus, any gases given out during distillation may be collected and preserved; but this is a circumstance, in pharmaceutical operations, which is attended to more with the view of guarding the operator against the effect of noxious gases, than of preserving gases for examination.
Rectification is the repeated distillation of any product obtained by distillation, when it is not perfectly pure. This second operation is carried on at a lower temperature, so that the more volatile parts only are raised, and pass over into the receiver, leaving the impurities behind. When the fluid is simply rendered stronger, as in the case of alcohol, by bringing over the spirit, and leaving behind the 'superfluous water, the operation is named dephlegmation or concentration. The process is called abstraction, when the liquid is distilled off from any substance; and cohobation, when the product is redistilled from the same materials, or from a fresh parcel of the same materials.
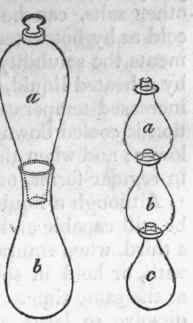
Sublimation is a species of distillation in which the product of the volatilization is condensed in a solid form; but as this condensation takes place at a higher temperature than that of a watery vapour, a much more simple apparatus is required. The process is conducted sometimes in a crucible with a cone of paper or another crucible inverted over it, in which the product is condensed; and as in this case it is light and spongy, it was formerly denominated flowers For other matters, which are less volatile, a cucurbit and capital, or a flask or phial, are employed, and sunk about two thirds in a sand bath. The aludel, a, a, b, b, c, which may be extended to any number of vessels, is generally used in this operation. The product in these cases is solid, and is denominated a sublimate.
b. Of the operations by which chymical changes are produced in the forms of bodies by the action of water, and other fluids.
When a solid body is thrown into a liquid and disappears, the transparency of the liquid remaining the same, the process is named solution; or solution is that operation by which the aggregation of a solid is overcome by a liquid, and a compound produced, which, retaining the fluid form, is transparent, and perfectly homogeneous. The liquid is generally supposed to be the substance exerting the active power, and has therefore been called the solvent or menstruum. It separates the particles of the solid or solvend from each other, and permanently suspends them by the state of combination into which they enter; but the attraction, as was before stated, is reciprocal, both as it regards the solid and the fluid. In general the solution of every solid in a liquid can be effected in a certain quantity only, or is limited; and when it is carried to its ultimate point, the liquid is said to be saturated. The solvent power, however, is not always limited, there being some instances in which a solid dissolves in a liquid in any proportion: thus gum and sugar dissolve in water in every proportion.
The solvent power of a fluid diminishes as it approaches to saturation, and the solution consequently goes on more slowly; but by raising the temperature, it proceeds again more rapidly, and a much larger portion of the solid is taken up than could have been dissolved at a lower temperature. This effect of temperature, however, does not take place in every instance; for chloride of sodium, for example, and some other salts, can be dissolved in nearly as great quantity by cold as by hot water. When an increase of temperature augments the solubility of bodies, a portion of the solid, taken up by a heated liquid, is retained in combination as long as the increased temperature exists, but separates again as the solution is cooled down to the temperature of the atmosphere, or lower; and when this is properly conducted, salts are obtained in regular forms, or crystallization takes place.
Although a liquid be saturated with one solid, yet it may be still capable of dissolving a portion of another, and even of a third, when saturated with the second; until it be combined with, or hold in solution, three, four, or five different bodies at the same time. The liquid, indeed, in this case does not dissolve so large a portion of any of the substances; but sometimes, from the mutual affinities which the substances exert, the whole proportion of solid matter dissolved is very much increased.
The solution of saline bodies in water requires no particular apparatus; as it can be conducted equally well in phials, or jars, or basins, provided the materials of which they are composed be such as can resist the action of the solvent.
Lixiviation is a term applied to solution when the saline body consists of both soluble and insoluble ingredients. On a great scale it is generally performed in large tubs or vats, having a hole near the bottom containing a wooden spigot and faucet. A layer of straw is placed at the bottom of the tub, over which the substance is spread, and covered by a cloth; after which, hot or cold water, according as the salt is more or less soluble, is poured on. The water, which soon takes up some of the soluble parts of the saline body, is after a little while drawn off by the spigot; and a fresh portion of water is successively added and drawn off until the whole of the soluble matter be dissolved. The straw in this operation acts as a filter; and the cloth prevents the water from making a hollow in the ingredients when it is poured on, by which it might escape without acting on the whole of the ingredients.
Continue to:


