Chapter XLI. Explosions - Fires And Other Accidents Produced By Ethereal Fluids Obtained In The Fabrication Of Fulminates -Treatment Of The Wounded
Description
This section is from the book "A Practical Treatise On The Fabrication Of Matches, Gun Cotton, Colored Fires And Fulminating Powders", by H. Dussauce. Also available from Amazon: A Practical Treatise on the Fabrication of Matches, Gun Cotton, Colored Fires and Fulminating Powder.
Chapter XLI. Explosions - Fires And Other Accidents Produced By Ethereal Fluids Obtained In The Fabrication Of Fulminates -Treatment Of The Wounded
When the necessary quantity of mercury is dissolved in the nitric acid, and the alcohol is added, we have seen the production of a lively reaction. The whole mass is strongly agitated, it disengages very abundant ethereal vapors, very inflammable, and have been often the cause of violent fires. At the moment that the mixture of nitrate of mercury, and alcohol produces the fulminate of mercury, the glass flask, in which the operation is conducted, may be broken, and the liquid fall into the fire; however, we think that this accident cannot result in any dangerous consequences, because the considerable quantity of dissolution will extinguish the fire, and the flame of alcohol inflame the powder with difficulty. But there are other dangers, more serious for the men, and which have their cause in the ethereal vapors. It is very important to give some details on this subject. Mr. Gaulthier de Claubry has devoted to it a long paper in the "Annales d' Hygiene Publique."*
This chemist has added to his paper an account of the accidents which he has himself passed through while preparing the fulminate. "For the past three years," says Mr. Gaulthier de Claubry, "the fulminates have been prepared in open retorts, the volatile products spread in the atmosphere, and the men engaged in that work have often complained of serious pains."
In the instructions annexed to his report, Mr. De Claubry observes that when the alcohol acts on the nitrate of mercury, it disengages a great quantity of vapor. The effect is very decided on the animal economy, but, at this epoch this action had not been studied, it was later that some dreadful accidents led to begin its examination. Indeed the volatile products we have spoken of, were then without use, but some manufacturers, to increase their profits, tried to employ them, and one of them obtained a patent to extract the alcohol from the ethereal fluids. This extraction has fur-nished alcohol in a quantity sufficiently large to introduce it in the trade. The process con-sisted in saturating with chalk the condensed liquid, and distilling the liquor separated from that product.
* October 8, 1839.
Mr. Gaulthier de Claubry, while visiting the establishment in which this operation was practised, learned from the manufacturer himself, that a man, while saturating the liquor with chalk, had been suffocated, and another trying to take him away had been nearly suffocated himself. The first had caused a great deal of uneasiness, on account of the violent nervous accidents which were manifested a short time after fainting away, and he was sick several hours. "I interrogated this man," says Mr. Gaulthier de Claubry, "to know the kind of pain that he experienced. He could not tell me anything but that he was taken immediately with a violent headache; that his strength left him; he cannot characterize the odor of the vapors.
"While there I made a saturation. The ethereal liquid was in a pan, the chalk was thrown into it and stirred with a long pole. The men kept away from the vapors, and advised me not to go near by. I examined carefully the odor disengaged, and that of hydro-cyanic acid was very manifest. I was immediately taken with a violent headache, which dissipated only after a night's rest."
These observations caused Mr. de Claubry to study more attentively the ethereal vapors we speak of. He ascertained that the products of the condensation on which they operate with chalk, have an agreeable smell of nitrous acid, and when they are inhaled for some time they occasion a painful sensation and headache.
Having treated with water the liquid product of the condensation, it separates a more or less considerable quantity of a yellowish liquid, having a strong odor of nitrous ether. Having tried to reunite a certain quantity with a little pipette, after two or three operations he experienced a whirling and a painful sensation of tightening in the head and the breast. "A drop of the liquid having touched my tongue," says he, "the pains became very strong, and the surrounding objects appeared to me as through a fog; a tingling in the ear added to these effects. After a few seconds a shivering of the limbs was manifested, and I experienced the beginning of fainting. A bottle full of ammonia was near me; I had strength enough to open it and smell it, which revived me.
"The pain in the breast was then very violent, that in the head insupportable; I perceived the surrounding objects, and I breathed with difficulty. I then left the laboratory. The cephalalgy continued for 24 hours, and I was well again after three days."
Strictly speaking, we can assimilate to those effects only those produced by the gaseous hydrocyanic acid. While, however, there are some differences in all the poisoning cases we have observed, we have never seen that pain at the posterior part of the head on which Mr. Gaulthier de Claubry insists.
The cephalalgy is often noticed, but the seat is not indicated. It is an important question to study.
Since the attention of the scientific world has been called to the effects produced by the inhalation of ether, it is very probable that the effects, observed in the manufacture of fulminates, are produced independently of the hydrocyanic acid, by vapors of ether.
Without doubt, nitrous ether, the odor of which predominates in those vapors, plays an important part in the production of the phenomena, for nearly all the effects mentioned by Mr. Gaulthier de Claubry have been observed as a sequel of the action of sulphuric ether.
What is the treatment to which the workmen, who experience the above accidents, are to be subjected?
It is evident that the precepts established by Orfila in cases of poisoning by hydrocyanic acid* must be followed. He says that the inspiration of a slightly ammoniacal water cures poisoning by this acid by stimulating the nervous system.
Mr. Simon has proposed chlorine as an antidote of hydrocyanic acid, and Orfila has shown that chlorinated water administered, even five minutes after poisoning, revives life.
In every case the action of fresh air is useful; it is then necessary to carry the person immediately to the open air, and far from the place of operation.
Happily, those accidents are now very rare, or rather are without examples. Nevertheless the dangers to which the workmen arc exposed have attracted the attention of chemists, and we owe to Mr. Chaudelon the construction of an apparatus destined to avoid them. +
* Annales d'Hygiene, vol. i.
+ Annales d'Hygiene, January, 1847, page 216.
This apparatus, represented below, is com-posed: -
Fig. 11.
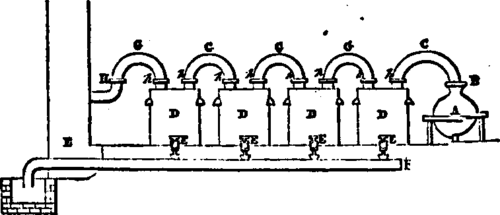
Fig. 12.
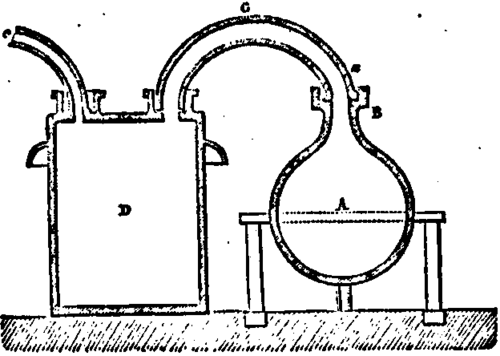
1st. Of two glass balloons, A of 10 gallons capacity resting on a frame, and in which are introduced the matters proper to prepare the fulminate of mercury. Each of these balloons has on the upper part of its neck a wooden collar B, covered with a sheet of lead, and adapting very tightly the collar by its circular groove a, forming a hydraulic fastening with the pipes C,C, which join the balloon to the apparatus of condensation. These tubes are supported by iron rods fixed in the floor of the room.
2d. Of a series of four earthen demijohns, D,D,D,D, provided at their lower part with a cork E, and carrying at their upper part tubu-latings, h,h,h,h, with hydraulic fastenings, in which adapt the earthen tubes G,G,G,G, which put all the demijohns in communication. Each demijohn has a capacity of about 22 gallons. The first has three tubulatings, the others have only two.
3d. Of an earthen pipe II, fixed in the wall, and carrying the deleterious vapors in the chimney.
4th. Of an earthen pipe F, fixed in the floor of the shop, and receiving from each cork the liquids condensed in the demijohns to conduct them in the saturating pan, which is outside of the room in the open air.
The apparatus being ready to work, pour in each tubulating h the necessary water to fasten; take out the pipe C, and introduce in one of the balloons 1 1/4 gallon of alcohol at 95°. Dissolve separately 12 ounces of mercury in 8 1/4 pounds of warm nitric acid at 36°, when the dissolution is achieved, and the temperature is at 170°, pour it into the balloon containing the alcohol. The pipe C being put back to its proper place, fill with water the groove of the collar, and leave the operation to itself. A few minutes after the reaction begins, and the vapors pass through the demijohns in which they condense. The portions which escape the condensation are carried away through the chimney.
During the operation the assistant has to pour from time to time cold water into each tubulating, to take the place of that which has 18 grown warm. When the reaction is achieved, take out the pipe C, being careful to shut immediately with a glass bell the tubulating led open, and proceed with the second balloon to the next operation. With the above proportions you obtain 20 ounces of damp fulminate, or 15 ounces when dry, and 3 1/2 quarts of condensed liquor.
Continue to:


