1. Sulphonations
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
1. Sulphonations
β-Naphthalene-monosulphonic Acid and β-Naphthol.
Reaction:

This product may be prepared by several different methods. If the β-monosulphonic acid is to serve for the preparation of β-Naphthol, the sulphuric acid must be completely utilized, as the product is so cheap that only the best process is capable of competing. (For further details, see β-Naphthol.) The method for preparing the Di- and Tri-sulphonic acids is given under H-acid.
The sulphonation of naphthalene at elevated temperatures (170o C.) leads to the formation of naphthalene β-sulphonic acid. A certain quantity of the alpha acid is always produced at the same time, amounting to about 15% at least, according to the results obtained by various experimenters. The researches of O. N. Wittl have shed a good deal of light, in certain directions, on the complex relationships involved. In actual practice, however, where it is necessary to obtain the highest possible yield of β-naphthol from the minimum possible quantity of sulphuric acid, Witt's process is hardly suitable.
1 Berichte, 1915, p. 743.
The following quantities give satisfactory results: -
260 gms. Naphthalene = 2 mols.
280 gms. Sulphuric acid, 66° Be. = 93 %.
The naphthalene used must be perfectly pure, must have no unpleasant tarry odour, and should not discolour on heating in a test-tube with concentrated sulphuric acid. The German tar-distilleries used to deliver naphthalene which met every requirement.
Laboratory Apparatus (see Fig. 2). - This consists of a sheet-iron, or, better, a cast-iron (or porcelain or glass), beaker of about 11 cm. diameter and some 20 cm. high. A well-fitting cover made of sheet lead is provided, through which pass the stirrer, thermometer, and tube for the addition of the acid. The type of stirrer shown in the figure has been found very suitable; it can be readily made from glass rod, and may be used for all purposes where the mechanical strain is not too great. In general, however, iron is to be preferred. The sleeve carrying the stirrer is best made of copper, as both glass and iron run very freely on copper on a small scale, and it does not tend to "corrode" the stirrer so much. The driving pulley may be made of bronze, as this metal also runs very well on copper, especially if vaseline be used as a lubricant. The thermometer should have the scale on the upper portion, and should dip down as far as possible. Further, I may remark that in all cases where it is necessary to watch the addition of any liquid very carefully, a dropping funnel with a drop-counter (as shown) should be used. The pot must stand on a good substantial retort-stand, and the copper "sleeve" must be fixed with a strong clamp. The thermometer and the dropping funnel must also be fixed firmly and in such a manner that the stirrer cannot come in contact with either.
260 gms. Naphth. 280 gms. H2So4.
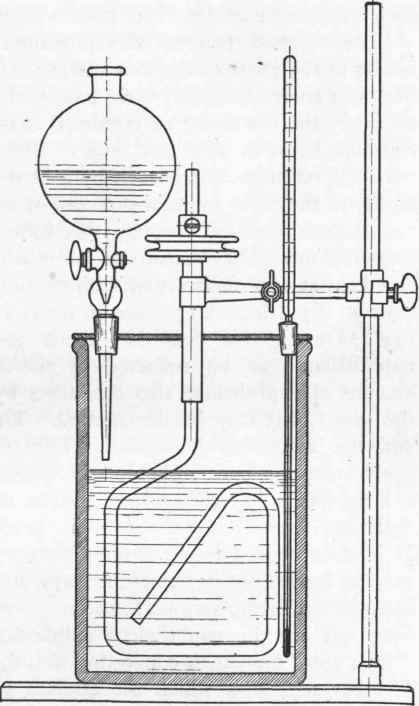
Fig. 2. - Sulphonating pot for naphthale sulphonic acids.
The weighed quantity of naphthalene is heated directly in the beaker to 165°, with continuous stirring. As soon as this temperature has been attained the sulphuric acid is allowed to run in during half an hour, the gas being so regulated that the temperature remains constant between 163o and 168o. The dropping funnel is then removed, its place being taken by a bent glass tube, which is fitted tightly to the cover by means of cork or asbestos paper. Water and naphthalene distil off through this tube during the course of the sulphonation. The mixture of naphthalene and sulphuric acid is now kept at 165o for an hour with continuous stirring, then for one hour at 167o, then at 170o for an hour, and, finally, for an hour at 173o. During this operation about 30 gms. water and 25 gms. naphthalene can be collected in the receiver. An appreciable amount of naphthalene also condenses by degrees on the cover of the vessel, but may be disregarded. The flame is then removed, and the apparatus dismantled. The resultant mixture contains, besides naphthalene sulphonic acid, a certain quantity of sulphones, a little free sulphuric acid, and some disulphonic acids, together with some resinous matters. The product should be colourless.
It is then poured into 1.8 litres water. The further working up may be carried out in numerous ways, and many different methods are adopted in the various factories. Some partially neutralize and then salt out the naphthalene sulphonic acids. Others prefer to "lime out" first, then converting into the sodium salt by means of Glauber salt, after which the calcium sulphate is filtered off, the residue being evaporated down and then worked up further. The simplest method is to salt out directly without attempting to neutralize at all, but this has the disadvantage that the strongly acid filtrate rapidly destroys both filter-cloths and filter-presses, and also that on drying the sodium salt of the monosulphonic acid the entire vicinity is polluted by the great quantities of hydrochloric acid which are given off.
1.8 1. Water.
The solution of the free sulphonic acids is now partially neutralized by sprinkling in 60 gms. of soda, with good stirring. 360 gms. of common salt are then added slowly; after a short time the liquid begins to solidify to a mass of large lumps which make further stirring very difficult. Nevertheless, the stirring must be continued until the mass again appears to be completely homogeneous, as only by this means can one ensure that the salt will be completely dissolved and that a precipitate will be obtained which will filter well. The actual amount of stirring required depends upon the speed of rotation of the stirrer, but in any case at least 6 hours will be requisite, otherwise the separation will be incomplete. The precipitate is then introduced into a suction filter provided with a cotton filter cloth and thoroughly pressed down. After removal from the filter the product is placed in a strong, moistened cotton cloth and pressed, gently at first, and then more energetically, in a screw press. The pressing should take at least 2 hours, otherwise too much mother-liquor remains in the cake. The hard mass is then ground up and dried completely at 100-1200 C.
Continue to:


