Benzidine From Nitrobenzene
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
Benzidine From Nitrobenzene
Reaction:

Phenylhydroxyl-amine.
Azoxybenzene.
Azobenzene.
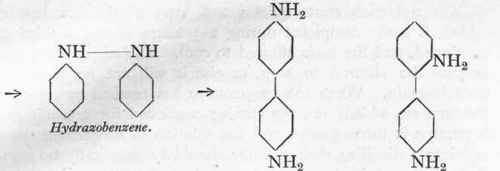
Benzidine {cm. 85 %).
Diphenyline. (ca.15 %).
Nitrobenzene is reduced to hydrazobenzene by means of cast-iron borings (in strong caustic soda solution), which must have the same properties as the iron used for the Bechamp-Brimmeyr reduction. It is very important to remove all oil from the turnings, or else too much will get into the benzidine. Further, the iron must be very finely divided as only the surface reacts. By the use of soda-lye, water, and iron turnings it is possible to reduce the nitrobenzene step by step and to obtain quantitative yields of hydrazobenzene, though the last stage is a delicate operation. Consequently, a modified process is often adopted as given in D. R. P. 138496; this will be referred to later.
The actual laboratory apparatus is illustrated on Plate XIV., Fig. 36. Since the iron turnings offer a considerable resistance to stirring, it is necessary to make use of much stronger apparatus than is usual in the laboratory. It will be found convenient to use a 1 h.p. water turbine or electric motor which can be made to turn a number of driving wheels. On the small scale the thermometer is best left out, owing to its resistance, measurement of the temperature of the oil-bath sufficing.
123 Gms. of nitrobenzene and 30 gms. of 60 % caustic soda solution are first placed in the reduction vessel; the mixture is then heated to 125o (oil-bath at about 140o). A reflux condenser is provided, as a certain amount of water distils off which carries away some nitrobenzene and reduction products. After the stirrer has been set in motion 400 gms. of very finely divided iron turnings are added during half an hour, which have been previously etched by means of 80 gms. of 60 % soda-lye at 1200. (The alkali attacks the iron with evolution of hydrogen which contains traces of strongly smelling phosphorus compounds; the etched iron looks like so much damp sand, and on exposing to the atmosphere readily cakes together to solid cement-like lumps, which, on a large scale, may lead to considerable difficulties.)
123 gms, Nitrobenzene. 30 gms. NaOH (60 %).
400 gms. Iron (or 500 gms. if necessary). 80 gms. NaOH (60 %).
The reduction starts quickly and, after all the iron has been added, is easily completed during 2-3 hours at 1250. Stirring is continued, and the mass allowed to cool. The stirrer must on no account be allowed to stop, or else it will not be possible to start it again. When the temperature has reached 750, 300 c.cs. benzene are added, and the stirring continued for 5 minutes, the apparatus is then opened, and the solution of azobenzene poured out into a distilling flask. There should be practically no iron in suspension as, with the concentration of caustic soda used, emulsions are rarely formed. The extraction is repeated three times at 75°, by which means the azobenzene will have been completely removed. Care must be taken to avoid the danger of fire.
The product may now be reduced directly to hydrazobenzene, but I do not recommend this procedure, as inseparable emulsions are almost always formed, i.e. the hydrazobenzene cannot be separated from the iron sludge. If, however, it is desired to use this method, which is that given in the patent referred to, then, instead of extracting, 300 c.cs. of benzene are added, and the temperature kept at 8o°. It is also necessary to add a further 50 gms. of caustic soda-lye (60 %), otherwise a hard cement is formed by degrees. To obtain complete reduction a further 200 gms. of iron turnings are added, the end of the reaction being indicated by the benzene solution becoming colourless. The separation of the hydrazobenzene is effected as with the azobenzene.
The azobenzene is obtained completely pure on distilling off the solvent, but before doing this it is necessary to remove all caustic lye by means of carbon dioxide and filtration. The yield is practically 100 % of theory or about 90 gms.
300 c.cs. Benzene.
300 c.cs. Benzene. 50 gms. NaOH (60 %).
200 gms. Fe
Continue to:


