Methylene Blue From Dimethylaniline
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
Methylene Blue From Dimethylaniline
The formation of Methylene Blue is of interest both from the scientific and technical standpoints, and will therefore be discussed before the actual methods of preparation are described.
Nitroso-dimethylaniline is prepared from dimethylaniline by treating the latter with sodium nitrite in hydrochloric acid solution. This nitroso compound is reduced, giving p-amino-dimethylaniline.
Reactions:
(a) p-Amino-dimethylaniline:

Dimethylaniline.
p - Nitroso-dimethylaniline.
p-Amino-dimethylaniline.
(b) The p-amino-dimethylaniline is oxidized in acid solution with a second molecule of dimethylaniline, and at the same time the thiosulphonic acid group is introduced into the molecule, which is effected by carrying out the oxidation in the presence of nascent thiosulphuric acid.
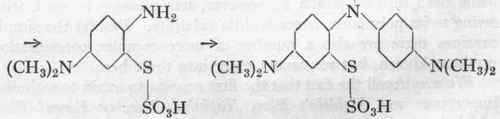
Thiosulphonic acid of p-amino-dimethylaniline.
Thiosulphonic acid of Bindschedler's Green.
(c) The thiosulphonic acid is now converted into Methylene Blue by closing the ring with the aid of more oxidizing agent:


(a) p-Amino-dimethylaniline.
24.2 Gms, (2/10 mol.) of pure dimethylaniline are dissolved in 75 gms. concentrated hydrochloric acid (30 %) and is then allowed to cool. The solution is treated with 150 gms. ice, and during 1 hour 14.7 gms. of 100 % sodium nitrite are run in as a 20 % solution, the nitrosation being complete in 4 hours. A further 110 gms. of 30 % hydrochloric acid are now added, together with 200 gms. ice, and 35 gms. good quality zinc dust are added during a quarter of an hour with mechanical stirring. The temperature may without danger be allowed to reach 25°. The solution is now colourless and neutral to Congo, and is filtered, the zinc dust being washed out with a very little water.
24.2 gms. Dimethyl-aniline. 75 gms.
30 %
Hc1. 150 gms. Ice.
14.7 gms. 100 % NaNo2. 110 gms. 30% Hc1.
35 gms. Zinc dust.
(b) Thiosulphonic Acid of Bindschedler's Green.
The oxidation at this stage must be effected in the presence of a zinc chloride solution which has no reducing action. Such a solution may be prepared by dissolving sheet-zinc in concentrated hydrochloric acid.1 The thiosulphuric acid is used in the form of aluminium thiosulphate, which is so strongly dissociated that it behaves like free thiosulphuric acid.
Before beginning the actual preparation of the Methylene Blue, solutions must be prepared of the necessary reagents, as an essential point in this operation is that the substances shall be added quickly and at the right temperature.
1 A method adopted in the works is to treat commercial zinc chloride liquor with sodium bichromate until there is no further reducing action; frequently 100-250 gms. bichromate are required for 100 kilos. of zinc solution.
Solution I = 38 gms. pure aluminium sulphate in 60 c.cs. water.
Solution II = 52.5 gms. crystallized sodium thiosulphate in 50 c.cs. water.
Solution III = 57 gms. sodium bichromate made up to 90 c.cs.
Solution IV = 20 gms. dimethylaniline in 27 gms. strong hydrochloric acid.
"Solution" V = 25 gms. very finely powdered pyrolusite (MnO2) made up into a homogeneous paste with 30 c.cs. water.
The clear neutral solution of p-amino-dimethylaniline is made mineral-acid with 4 gms. concentrated sulphuric acid and 100 gms. of 50 % non-reducing zinc chloride solution is added. The beaker is placed on a felt pad and heated up by blowing in steam. Solution I is added at the ordinary temperature with good stirring, then Solution II, and after 2 seconds one-third of Solution III,corresponding to 19 gms. of sodium bichromate. By passing in dry steam the temperature is raised to 400 in one minute, Solution IV is added, then the remainder of Solution III, and the whole heated rapidly to 700. The liquid becomes dark greenish-blue, owing to the formation of the thiosulphonic acid of Bindschedler's Green. As soon as 700 is attained, suspension V is added, and the whole heated to 85°.
The reason for adding the manganese dioxide is to convert the sulphurous acid which is set free during the ring-formation into the harmless dithionate. In place of the pyrolusite, 40 gms. copper sulphate may be used with equal success, the cupric salt being converted into the insoluble cuprous salt.
At 85° the solution develops a fine bronzed appearance and the resultant dye is precipitated from the concentrated zinc chloride solution. After half an hour the mixture is allowed to cool to 500, and 70 gms. concentrated sulphuric acid are added, which dissolves up the manganese salt, aluminium hydroxide, and chromic oxide. The product is filtered off at 200 and washed with a little 10 % brine. The crude blue is dissolved in a litre of water at 100°, filtered from insoluble matter, and the clear filtrate salted out with 50 gms. of ordinary 50 % zinc chloride solution and 150 gms. common salt. After 24 hours, the zinc chloride double salt comes out as a fine red bronzed precipitate which is filtered off and washed with a little 10 % salt solution; it is then dried at a temperature not exceeding 500, a yield being obtained of about 44 gms. pure concentrated colour.
4 gms. 100 %
H2So4.
100 gms.
50% ZnCl2.
38 gms. A12(So4)3 + 18H2O.
52.5 gms.
Na2S2O3
+5H2O.
19 gms. Na2Cr2O7.
38 gms. Na2Cr2O7. 25 gms. MnO, (about 88%).
70 gms. H2So4, 66° Be.
1 litre H2O.
50 gms. ZnCl„ 50 %. 150 gms. NaCl.
Notes on Works Technique and Practice. - The method described above was originated by Bernthsen and Ulrich, who also recommended the use of aluminium thiosulphate. The use of pyrolusite or copper sulphate is general. Large quantities are not dealt with at one time as quick heating up is essential. On the large scale the finished dye is usually filtered off in frame-filters (see Plate VI.), and after draining, is put into small bags and centrifuged.
Continue to:


