Reduction To Hydrazobenzene. Continued
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
Reduction To Hydrazobenzene. Continued
55 gms. H2So4, 66° B6.
50 gms. Na2Co3.
The apparatus used on the works must be built very strongly as the stiff iron mass offers very great resistance to stirring. Plate XL, Fig. 29, illustrates a reaction vessel of this type with duplex stirring gear. In this case the stirrer, however, is made somewhat differently, like a plough, in order that the paddles may go through the iron sludge more easily.
The extraction may be carried out in the reduction vessel itself, the benzene solution being run off through side outlets. Special extraction apparatuses are made also, in which the iron, after separating from the reduction liquor, is extracted. Tip-up vessels, which can be quickly and easily emptied, are sometimes employed. Apparatus of this type is very heavy, but has the advantage over those which are emptied through a bottom exit-tube that it has no spiral conveyor which easily gets stopped up.
Increasing use is being made of centrifuges for separating the mother-liquors from the precipitates, with the exception of the filtration of the benzidine sulphate, which is frequently performed by means of a nutsch (Fig. VI.) or a filter-press. So far no use has been found for the diphenyline obtained as a by-product. Although the 5 % loss on distillation has deterred most factories from isolating the benzidine in this way, I am quite certain, from my own experience, that this loss is apparent rather than real.
The superiority of the distilled benzidine shows itself particularly in the manufacture of complex triazo colours, as in these cases the increased price is more than made up for by the better yields. Colours such as Direct Deep Black E.W. (q.v.) or Dianil Brown 3GN, when prepared from quite pure components, will always excel those obtained from less pure materials. It is perhaps hardly necessary to remark that all by-products, such as caustic-lye, unused iron, and solvent should be most carefully recovered.
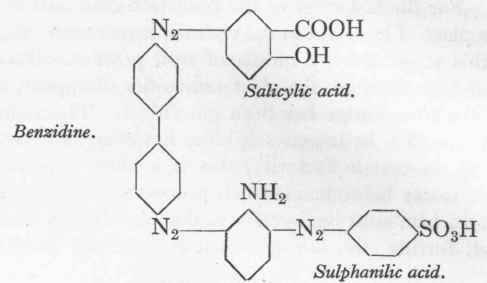
m-Phenyline diamine.
Dianil Brown 3GN.
Finally, I may mention that the second process, the electrolytic, will, in my opinion, gradually displace even the Weiler-ter-Meer process, in spite of all difficulties. It has a great superiority in that absolutely no metal is required, which was a great advantage during the war, when it was extremely difficult to obtain cheap zinc dust. At the present time there is only one factory, and that in Switzerland, which carries out this process successfully.
2:2'-Benzidine Disulphonic Acid from Nitrobenzene.
Reaction:

Phenylhydroxylamine sulphonic acid.
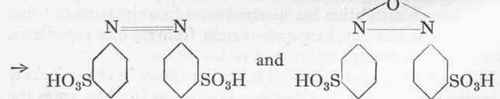
Azobenzene disulphonic acid.

Hydrazobenzene-disulphonic acid.
2:2'-Benzidine disulphonic acid.

Fig. 14. - Stone vacuum filter for strongly acid precipitates (scale 1:30). 1. Vacuum connection. 2. Outlet tube for filtrate. 3. Air by-pass. 4. Connection for emptying out into waste tanks or drains.

Fig . 15. - Transportable frame-filter for coarsely crystalline precipitates (scale 1 : 25.)
Plate VI.
The preparation of nitrobenzene sulphonic acid has been fully described in connection with metanilic acid. The reduction process differs only from similar reactions in that it is carried out purposely in dilute aqueous solution in the present case, and in three stages. It is possible to obtain benzidine disulphonic acid with the use of a minimum quantity of caustic soda and zinc dust.
If the sodium salt used is not quite pure, the press-cakes of sodium nitrobenzene sulphonate corresponding to 100 gms. nitrobenzene are dissolved in about 30 gms. soda so that the solution is exactly neutral to litmus; it is then made up to 1 1/2 litres at 10°, 10 gms. of ammonium chloride are added, and the liquid vigorously stirred by means of a propeller stirrer. 120 Gms. zinc dust are then added during 2 minutes, a teaspoonful at a time; finely crushed ice is also added from time to time to keep the temperature below 200, stirring being continued for 20 minutes. 120 Gms. 30 % caustic soda-lye are poured in quickly, and the mass warmed up to 700 without stirring. The solution, which was originally colourless, at once becomes orange-yellow owing to the formation of the azo- and azoxy-benzene sulphonic acids, and is allowed to stand for at least 3 hours or, better, over-night.
Next day the product is neutralized cautiously by the addition of about 90 gms. concentrated hydrochloric acid, drop' by drop, until no reaction is given with thiazole paper. After the liquid has been heated up to 8o° a further 40 gms. zinc dust are added. If the colour has not disappeared after 5 minutes, a further quantity of hydrochloric acid is dropped in slowly at 75-800. The change of colour from a dirty brown to a clear light grey takes place in less than 5 seconds as soon as the neutral point has been reached. The liquid now contains the hydrazine sulphonic acid, its volume being about 1.8 litres, and is quickly run through a filter to prevent further reduction to metanilic acid, the zinc dust being well washed out. After cooling, the filtrate is treated at 200 with 120 c.cs. conc, hydrochloric acid. In a few minutes a glistening precipitate forms, consisting of colourless hard crystals of 2:2'-benzidine disulphonic acid, the liquor becoming yellow through autoxidation; a few drops of stannous chloride solution are therefore added to decolourize it. Although the benzidine disulphonic acid is extremely sparingly soluble in water (less than 1 gm. per litre), it separates out very slowly, so that the product must be allowed to stand for a couple of days before filtering and washing with cold water. Yield about 65 gms.
Notes on Works Technique and Practice. - On the large scale,
100 gms. Nitrobenzene as sulphonic acid. 1500 c.cs. Nh4c1.
120 gms. Zinc dust.
120 gms. NaOH (30 %)• ca. 90 gms. conc. Hc1.
40 gms. Zinc dust.
120 c.cs. conc. Hc1.
where one has to deal with volumes of 4000 to 5000 litres, the crystallization of the 2:2'-benzidine disulphonic acid occupies at least 3 days. In order to secure as rapid cooling as possible a leaden coil, through which cold water is circulated, is placed in the wooden tub.
Owing to its insolubility the sulphonic acid must be diazotized indirectly; it is dissolved in the requisite amount of water and soda, the neutral solution mixed with sodium nitrite, and the mixture allowed to run in a thin stream into hydrochloric or sulphuric acid.
Continue to:


