Chapter XXVII. Distillation Of Mixtures Of Water And Ethyl Alcohol, With The Other Volatile Products Of Fermentation
Description
This section is from the book "Distillation Principles And Processes", by Sydney Young. Also available from Amazon: Distillation Principles And Processes.
Chapter XXVII. Distillation Of Mixtures Of Water And Ethyl Alcohol, With The Other Volatile Products Of Fermentation
The problem of how to separate the ethyl alcohol from the water has already been dealt with. The rectification or removal of the volatile by-products remains to be considered. The majority of these sub-stances are produced in the fermentation, but some of them are formed by chemical changes taking place during the process of distillation. To the latter class belong certain esters produced by the interaction of acids and alcohols present in the fermented wash. The elimination of these acids in the early stages of rectification will reduce the amount of esters produced.
The volatile by-products include the following substances :1 Aldehydes. n-Propyl alcohol. Iso-propyl alcohol. n-Butyl alcohol. Iso-butyl alcohol. Iso-amyl alcohol. d-Amyl alcohol. Hexyl and heptyl alcohols. Furfurol bases. Iso-butyl glycol.
Acetal.
Glycerine.
Fatty acids, including formic acid. Ethyl acetate and formate. Ethyl butyrate. Ethyl iso-valerate. Iso-amyl acetate. Iso-amyl iso-valerate. Terpenes.
The by-products can be roughly divided into two classes : (1) "head products," which tend to pass over into the distillate more readily than ethyl alcohol, and (2) "tail products," which do not do so. But no hard-and-fast distinction can be drawn, as some of the by-products pass from class 1 to class 2 as the composition of the liquid to be distilled varies.
The head products are more readily removed from dilute alcoholic solution (fermented wash) than from concentrated alcoholic solutions which have not received a preliminary purification. In the initial stages of the heating of the fermented wash the liquor is practically subjected to a process of distillation in a current of steam. Owing to the relatively small proportion of impurities present, the first fractions of the distillate will contain the greater proportion of the impurities (fusel oil) contained in the original fermented wash. The distillation constants of the ester impurities are also high, and these bodies will also come over in the first fraction.
1 Harden, Alcoholic Fermentation, 1914, p. 85; Maercker Delbruck, Spiritus-Fabrikation, 1908, p. 761 ; Windisch, Arb. Kais. Gesund., 1892, 8, 228.
Distillation of Mixtures of Alcohol and Water
In the distillation of mixtures of liquids four groups are usually considered.
(a) Non-miscible liquids.
(b) Partly miscible liquids.
(c) Closely related liquids - miscible in all proportions.
(d) Liquids not closely related but miscible in all proportions.
As the mixture of ethyl alcohol and water will come in group d, the remaining groups (a, b, and c) will only be briefly referred to here - their more detailed study has been considered earlier in this volume.
With non-miscible liquids (group a) the boiling point of each individual liquid in the mixture depends on the partial pressure of its own vapour and generally is not influenced by the other substances present. Since the constituents of these mixtures may be considered as independent of each other the laws of distillation are simple and the theoretical calculated results obtained from the known vapour pressures agree very closely with those found experimentally. These laws have been developed in the main by Naumann,1 Pierre and Puchot,2 Brown.3
In the working up of the by-products from fermentation liquors sometimes two layer mixtures are formed - e.g. some higher alcohols and water. These mixtures would be of the type included under group b.
In mixtures of liquids included in group c, the relation between the vapour pressure and molecular composition may be represented by a straight line, and for such a mixture as chlorobenzene and bromo-benzene the relation holds accurately.4 The relation between the composition of the liquid mixture and that of the vapour evolved from it according to Brown's formula may be expressed as follows :where m'A and m'B are the relative masses of the two substances in the vapour, mA and mB their relative masses in the liquid mixture, and PA and PB the vapour pressures of the pure substances at the boiling point of the mixture. Substituting a constant c for the ratio PA/PB, a better agreement was obtained between the calculated and observed results. As shown earlier in this volume, Brown's experimental results do not agree well with this formula, and the experimental evidence available points to the conclusion that the formula is only applicable to these liquid mixtures of which the relation P = mPa +(1 -m)Pb holds good, P being the vapour pressure of the mixture and PA and PB the vapour pressure of the two pure substances at the same temperature, and M the molar fraction of the substance A.1
1 Ber., 1879, 10, 1421, 1877. 2 Compt. rend., 73, 599, 74, 224.

3 Tram. Chem. Soc, 1879, 37, 547.
4 Cf. Brown, Trans. Chem. Soc., 1880, 35, 541, 37, 49 ; 1881, 39, 304 and 517 ; Young and Fortey, Trans. Chem. Soc., 1903, 81, 768, 902, and 83, 45, etc.
Duhem and Margules independently suggested the formula for the relation between the molar composition of the liquid mixture and the partial pressures PA and PB of the components in the vapour, and the formulae of Lehfeldt and of Zawidski are based on this equation. Doubt has been thrown on the graphic method of Margules (which relies upon the measurement of the slope of the total pressure curve at its two ends) by the subsequent work of Rosanoff and co-workers.2 These latter have developed a general law which may be expressed as


The alcohols may be considered as being formed from water by the substitution of an alkyl group for one of the hydrogen atoms. They may also be considered as hydroxyl derivatives of the saturated paraffins. From both of these aspects the subject has been studied by Young3 in his investigation of the relationship of the alcohols to water on the one hand and to the paraffins on the other hand. In general the influence of the OH group diminishes and that of the alkyl group increases as we pass from water up the series of alcohols. The vapour pressures of mixtures of methyl alcohol and water are always intermediate between those of the pure components, and the curve representing the relation between the vapour pressure and the molecular percentage of methyl alcohol deviates only slightly from a straight line. The maximum differences between the pressures represented by the curve and a straight line joining the two ends of the curve is 43 mm. on a certain scale. On the same scale the maximum difference for mixtures of ethyl alcohol and water mixtures is 315 mm.4 For further study of this relationship of the alcohols see earlier sections in this book. The behaviour of methyl alcohol and water mixtures appears to place these mixtures in group c rather than group d, but with ethyl alcohol and water mixtures there is a closer connection with group d. With the higher soluble alcohols, such as the butyl alcohols or the amyl alcohols, the connection with group d is very complete within the limits of solubilitv, and if the formula used for non-miscible or closelv related miscible liquids is applied large deviations from the calculated results are obtained.
1 Of. Young, Proc. Royal Dublin Soc, 1920, 15, 47.
2 J. Amer. Chem. Soc., 1911-1920.
3 Trans. Chem. Soc., 1902, 81, 707.
4 Konowalow, Wied. Ann., 1881, 14, 34.
The study of the vapour pressures of mixtures of group d was undertaken early in the nineteenth century by Gay-Lussac (1815), Boit (1816), Magnus (1836), Begnault (1853), and later by Berthelot, Young, and others. It is, however, to Duclaux1 that we are indebted for the first co-relation of the earlier work on the study of the distillation of dilute solutions of certain substances (such as alcohols or fatty acids) and for the establishment of a relation between the amounts of the constituents in the mixture to be distilled and their amounts in the vapours.
Distillation of Dilute Aqueous Solutions of Substances
Volatile in Steam
Duclaux originally studied the rates of distillation from the analytical standpoint. He distilled a known volume of a dilute solution of the volatile acid or alcohol and collected the distillate in several equal fractions. By expressing the amount of acid or alcohol in each fraction as a percentage of the total amount distilled, he was able to obtain a series of constants which served to identify the particular acid or alcohol. For mixtures of two acids or two alcohols each substance approximately follows its own law of distillation. The same line of reasoning applies in the case of either alcohols or acids, and as the distillation of the volatile acids was first studied a typical example of the distillation of dilute acid solution will be considered.
A solution containing at the most 1-2 grams of acid is made up to 110 c.c. and distilled in a flask of 200-300 c.c. capacity, using an ordinary Liebig condenser. Ten successive fractions of 10 c.c. each are collected. Each fraction is titrated in turn, so that the acid in all the 10 fractions can be determined. The titration of each fraction is determined as a percentage of the total titration for 100 c.c. of distillate.
Suppose in the distillation of a solution of a volatile acid the following values are obtained for titration of each fraction :-
Total. | Percentage. | ||
1. | 4.l c.c. | 4.1 c.c. | 155 |
2. | 35 „ | 7.6 „ | 28.8 |
3. | 3.1 „ | 107 ,, | 40.6 |
4. | 2.85 „ | 13.55 „ | 513 |
5. | 2.65 „ | 16.2 „ | 614 |
6. | 245 ,, | 18.65 „ | 70.6 |
7. | 2.2 „ | 20.85 „ | 79.0 |
8. | 2.1 „ | 22.95 „ | 86.9 |
9. | 1.75 „ | 24.7 „ | 936 |
10. | 1.7 „ | 26.4 „ | 100.0 |
Total titration for 100 c.c } | 26.4 „ |
On this principle tables have been constructed showing the percentage of acid in all fractions, calculated on the amount distilled in 100 c.c. of distillate.
The following table gives these values obtained by Duclaux :1 Ann. de phys. et chim., 1878, and later.
Distillate. | Acetic acid. | Propionic acid. | n-Butyric acid. |
20 c.c. | 15.2 | 24.0 | 33.6 |
30 „ | 23.4 | 35.3 | 47.5 |
40 „ | 32.0 | 46.2 | 60.0 |
50 „ | 40.9 | 56.8 | 70.6 |
60 „ | 50.5 | 66.7 | 79.5 |
70 „ | 60.9 | 76.2 | 86.5 |
80 „ | 71.9 | 85.0 | 92.5 |
90 „ | 84.4 | 93.0 | 97.0 |
For mixtures of two acids, Duclaux states that each acid follows its own laws of distillation. This is not strictly true, but the error involved is generally less than the experimental error.
It follows that if a mixture containing two volatile acids is distilled, the distillation values will lie between those of the two acids, and will be a measure of the composition of the mixture. This can be seen in the following example. A mixture of 1 .25 molecular proportion of butyric acid and 1 of acetic gave the following results of distillation by Duclaux's method: -
Percentage. | |
30 c.c. | 36.7 |
40 „ | 47.6 |
50 „ | 57.4 |
60 „ | 66.7 |
70 „ | 75.2 |
80 „ | 82.4 |
For 30 c.c. of distillate the corresponding values for acetic and butyric acids are 23 4 and 47.5 respectively. If the acid is originally supposed to be butyric acid to which increasing amounts of acetic acid have been added, the difference between the value for butyric acid figures and the corresponding experimentally derived value will be a measure of the acetic acid in the mixture. Thus the molecular proportion of acetic acid present in the solution is 47.5-36-7/47.5-23.4 = 0.448.
Similarly for fraction 40 c.c. the molecular proportion of acetic acid present
= 60.0-47.6 / 60.0-32.0 = 0.443 for 60 c.c.
79.5-66.7 / 79.5-50.5 = 0.442
The method of calculation can be expressed by the general formulaC1-C3 / C1-C2 = mol. ratio of lower acid in the mixture ; where C1 = distillation constant of acid 1 having higher distillation constant, C2 = distillation constant of acid 2 having lower distillation constant, C3 = distillation constant of mixture of 1 and 2.
Similarly the molecular ratio of the acid of high molecular weight is given by general expression mol. ratio of acid of high molecular weight.

For mixtures of unknown acids, the analysis is somewhat tedious. Calculations must be made for trial mixtures until a certain mixture gives constant results for the proportions of acids present. The calculations are simplified by constructing tables showing the results which would be expected by mixtures of known composition.
While the original method outlined by Duclaux did not give concordant results with different workers, a large amount of experimental work has been carried out to remedy this defect. It has been shown that loss of heat from the exposed flask and still-head, by convection currents and radiation, leads to irregular results. Various arrangements for preventing this have been used. It is advisable to keep the exposed flask at no higher temperature than that of the vapour, otherwise errors will be introduced by splashing and complete evaporation of drops of solution. The best conditions for preventing condensation are obtained by surrounding the flask with a steam jacket.
In the case of acids the acidity of the distillate may be determined by simple titration, but with alcohols the method of estimation is not so direct. The "drop method" used by Duclaux1 depends on the fact that the surface tension of different mixtures of alcohols and water tends to diminish with the increased percentage of alcohol.
The Coefficients of Duclaux, Sorel, and Barbet. - The original formula of Duclaux may be expressed thus :where a and b represent the percentage by volume of alcohol and water respectively in the original liquid, and da and db the percentages of alcohol and water respectively in the vapour. The above relation may be represented by a hyperbola. The value of c with dilute solutions of various alcohols varies from 10.9 for methyl alcohol to 50 for amyl alcohol and 61 for capryl alcohol. The coefficient c increases with the molecular weight of the alcohol, and the higher alcohols pass over on distillation of their aqueous solutions more readily than do the lower alcohols.

In Chapter XXVI. reference is made to Groning's figures for the distribution of alcohol-water mixtures. It was from a study of these figures that Sorel was led to his investigation on the distillation of alcohol and water mixtures.2 He used a copper retort of 5 litres capacity submerged in a bath of glycerine for the heating of 4 litres of liquid. The distillate was collected in about 40 equal fractions, and the alcohol content in each fraction estimated by a density determination. If the volumes are plotted as abscissae and the concentrations of the remaining liquid as ordinates, we have what Sorel calls the curve of purification of the original liquid. If V is the volume remaining at any particular moment, a the Gay-Lussac concentration of the liquid, i.e. the percentage by volume of the alcohol in the mixture, and U the Gay-Lussac concentration of the liquid distilled, i.e. the percentage by volume of alcohol in the fraction distilled; then at any moment
1 Ann. Inst. Pasteur, 1895, 9, 575. 2 Comptes rendus, 1892.
Va = (V-dV)(a-da) + dVU,
da or U=a+ ___ dv
The value of U is obtained from the curve of purification by finding the angular coefficient of the tangent.
Mariller1 makes special use of a constant K, usually called the coefficient of solubility, or, better, the coefficient of enrichment, which is given by the following equation: or in above notation -


With dilute alcohol solution (1 per cent) the enrichment coefficient K on Sorel's calculations is 9 .9 for ethyl alcohol at 1 per cent. concentration, and this figure gradually diminishes as a increases. With a = 50, K = 1 .5 ; between a = 50 and a = 95.47, K falls from 1 .3 to 1. The corresponding values of K from Duclaux's and Groning's figures are nearly equal, but somewhat higher figures are obtained from Sorel's data. Although Duclaux mainly dealt with the problem of dilute alcoholic distillation from the analytical standpoint, both Sorel and Barbet (especially the latter) were concerned with the industrial problem of the distillation of fermentation liquors. If S is the weight of the impurities present in 1 kilogram of the mixed vapours from fermentation liquors, and "s" the weight of impurities in 1 kilogram of the original liquor, then according to Sorel S = K1s + K2s2 + Kss3 +. . . .
If "s" is considered to be small then S = K1S.
The distillation of mixtures of ethyl alcohol, higher alcohols, various esters, aldehydes, etc., and water over a wide range of alcohol concentrations has been made by Barbet (La Rectification et les colonnes rectificatrices, 1895, p. 46).2
1 Le Bulletin de l'Association des Chimistes, 1911, 473-490, and La Distillation fraclionnee, p. 23.
2 See also Sorel, Comptes rendus, 1894, 118, 1213; Sorel, La Rectification de l'alcool, 1894, pp. 18.33; Sorel, Societe d'encouragement, Bulletin de mai, 1891, pp. 226, 240.
The ratio percentage of "impurities " in ethyl alcohol in distillate percentage of " impurities " in ethyl alcohol in liquid he designated by K', and it is known as the coefficient of purification.
If K= percentage by wt. of impurities {e.g. amyl alcohol, etc.) in vapour percentage by wt. of impurities (amyl alcohol, etc.) in liquid then K'= k'
In other words, K' indicates how far distillation will remove this impurity from the alcohol apart altogether from the quantity of water present. This "coefficient of purification ' is of a more practical value than the coefficient of enrichment. The experimental methods of Barbet are also more closely related to the methods employed in the distillery than, are those of Sorel.
Fig. 102 gives the value of K 'for various impurities found in fermented liquid. At the point where K' = l there is a change of direction in the course of the purification. So long as K'>1 the alcoholic vapour is richer in the impurity than the alcohol in the original liquid. When K' = 1 the concentration of impurity is the same in liquid and vapour. Finally, when K'< 1 the impurity tends to become concentrated in the liquid.
It should be noted here that the amount of impurity present in the original liquid as examined by Sorel never exceeded 2 per cent. In practice the concentration of some of the impurities considerably exceeds 2 per cent at certain stages of the rectification. Their behaviour will depend largely on their vapour pressures under the varying conditions. In the case of such an impurity as amyl alcohol the formation of a mixture of maximum vapour pressure with water will greatly influence the result. In practice there is always more than one impurity present, and this will to some extent alter the values of K'. In all the cases shown the value of K' falls with rising alcoholic content of the liquid. This is due partly to the fact that, as the alcoholic strength rises, the temperature of ebullition of the liquid falls, and with it the vapour pressure of the impurity. Another factor affecting the result in the same direction is the great solubility of all the impurities in alcohol and the very slight solubility of most of them in water. With the exception of propyl alcohol and aldehydic bodies, the greater part of the impurities are insoluble or only partially soluble in water.
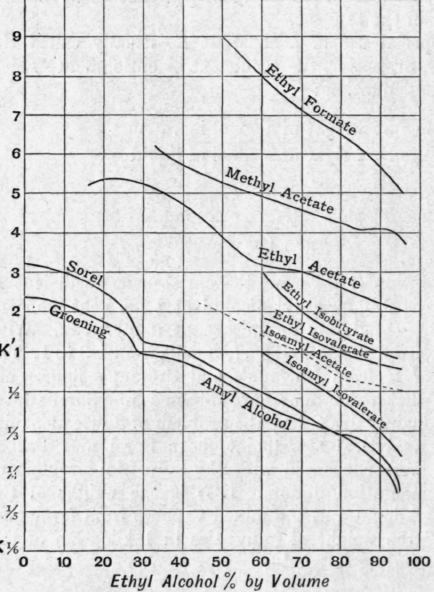
Fig. 102.
The fact that the tail products are more readily removed from dilute alcoholic solutions than from more concentrated solutions is utilised in at least one type of modern still. According to the patent of E. Guillaume1 the alcoholic liquors to be purified are diluted if necessary in such a way that the products considered as tail products, e.g. amyl alcohol, may behave during the distillation like head products relative to the mixture of ethyl alcohol and water. These liquors are then fractionally distilled in a continuous distilling column of sufficient power and number of plates to enable a large part of the products hitherto considered as tail products to be removed at the same time as the so-called head products, so that to the bottom of this distilling column there comes only a practically pure mixture of water and ethyl alcohol.
Distillation of Aqueous Alcohol Solution at Constant Volume
The work of Duclaux, Sorel, and Barbet deals with the distillation of alcoholic solutions at varying volumes. A brief consideration will be given to the distillation of aqueous solutions of volatile substances at constant volume.2
As a result of the distillation of aqueous phenol solutions at constant volume, Naumann and Muller3 came to the conclusion that under constant conditions of temperature and pressure, the amount of sub-stance distilling was proportional to the amount of substance in the flask. They deduced values for the ratio of the amount of substance in the flask to the amount distilling in each fraction, and also for the ratio of the titration or value of a fraction to the one preceding it. Stein 4 distilled aqueous solutions of certain volatile substances in a current of steam, and observed certain regularities in the distillation. To express the rates of distillation, values were calculated from the formula
1 a - log v a-x
1 Eng. Pat. 5794/1902.
2 Cf. Reilly and Hickinbottom, Proc. Royal Dublin Soc, XV. 37, 514.
3 Berichte, 1901, 34, 224. 4 J. pr. Chem., 1913, 88, 83.
U where v = volume distilled, a = amount of substance originally present, x = amount of substance distilled.
According to Nernst's law of distribution
The proportion of constituents in the distillate are assumed to be the same as the ratio in the vapour phase in a state of equilibrium. This assumption is probably not strictly accurate, but approaches accuracy if the distillation is carried out slowly and regularly.
Let x = the amount of alcohol in distillate after volume v has been distilled, initial amount of alcohol in distillation vessel, density of water vapour in distillation vessel, weight of water per unit volume of distillate, then if dx is the quantity of alcohol coming over in a quantity dv of distillate, concentration in vapour phase approximately, concentration in liquid phase approximately, where V = constant volume of liquid in a flask, then which gives or writing



Then A is a constant, assuming is constant.
It may be readily seen that the coefficient K used on p. 287 is related to the distillation constant (A) given above as follows: -
The distillation constant (A) varies to some extent with the concentration of the alcohol (of. distillation constants in the distillation of 0.8 per cent and 4.8 per cent ethyl alcohol solutions). The values for A can strictly be compared only with the values of K for dilute solutions. Variations in the temperature of the aqueous alcoholic solution may partly account for the alteration in the distillation constant.
Another disturbing effect may be due to association of the alcohol. Murray1 has adduced evidence in favour of the view that the molecules are associated in the liquid state but are not generally associated in aqueous solution. If there still exist in the solution some associated molecules at the concentration employed, there will be a continual change in the state of aggregation as the distillation proceeds. Under these conditions Nernst's law of distribution will only hold approximately. The available evidence, however, mainly favours the view that if there is any combination between the alcohol and water it must be very slight.2
The above treatment only deals with the case of a single alcohol in solution. The theory of mixtures may be considered as follows : - Let a and b be the amounts of each alcohol present in the distillation vessel initially and xn and yn the amounts distilled over in n-fractions.
We get for first alcohol, for second alcohol, or
The fraction of original alcohols left write m and n represent the ratio in which alcohols were present initially, and we have m + n = l,

We have then writing if we write p'n = per cent of 1st alcohol coming over in n-fractions,

,, ,, P" ,, = ,, ,, 2nd ,, ,, ,,

When a mixture of alcohols in dilute solution is distilled, each alcohol will distil at a rate independent of the other alcohols present but depending on its own concentration.
1 Amer. Chem. Journ., 1903, 30, 193.
2 Reilly and Hickinbottom, Proc. Roy. Dublin Soc., 1921.
The distillation constant (A) of several of the lower alcohols have been determined at constant volumes.1 The experimental results are given in Table 111.
It may be seen that the distillation constants of the alcohols increase in an approximately regular manner with an increase in the molecular weight. The influence of other impurities such as aldehyde, esters, etc., is not considered as the quantity of these present is much less than that of the higher alcohols. Their distillation constants, however, may be calculated from the ceofficient given in Table 111.
Continue to:


