Composition Of Liquid And Vapour Phases. Exp Det. Part 2
Description
This section is from the book "Distillation Principles And Processes", by Sydney Young. Also available from Amazon: Distillation Principles And Processes.
Composition Of Liquid And Vapour Phases. Exp Det. Part 2
Lehfeldt's Apparatus
Lehfeldt's apparatus1 for distillation at constant temperature is shown in Fig. 25. The still, which is in the form of a large test tube, is provided with a cork perforated with two holes, through one of which passes a thermometer, the bulb of which is covered with a little cotton-wool, which dips just below the surface of the liquid. Through the other hole passes the delivery tube, E, E, connected with the condenser F, which is provided with a tap, G, for drawing off the distillate, and a tube connected with a pressure gauge, air reservoir and air pump. The bell-jar, J, contains either cold water or a freezing mixture. The still is heated by water in a large beaker placed on a sand bath ; the water is constantly stirred and its temperature is registered by a thermometer. In order to prevent back condensation in the vertical part of the delivery tube, an incandescent electric lamp, with the ordinary conical shade, is lowered as close as possible to the water bath, and a cloth is hung round the whole ; the top of the apparatus is thus kept at least as hot as the bath.
1 Loc. cit.
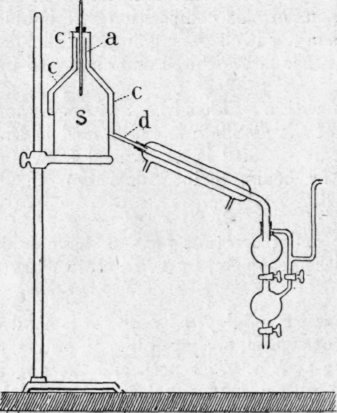
Fig. 24. - Brown's apparatus.
The quantities of material employed by Lehfeldt were small; about 30 c.c. of the mixture to be investigated were placed in the still and three fractions of about 1 c.c. each were usually collected and examined separately by means of a Pulfrich refractometer.

Fig. 25. - Lehfeldt's apparatus.
Steady ebullition was ensured by placing a piece of pumice stone, weighted with copper wire, in the still, and the pressure was adjusted from time to time to keep the boiling point as nearly constant as possible.
Preliminary determinations of the refractive powers of mixtures of the liquids investigated were made.
Zawidski's Apparatus
Zawidski1 employed an apparatus which is similar in principle, but more elaborate than Lehfeldt's ; it is shown in Fig. 26. The still, a, of about 200 c.c. capacity, and containing in each experiment from 100 to 120 c.c. of liquid, is heated by a water bath, G, provided with a stirrer and thermostat. Back condensation is prevented by coiling copper wire round the upper part of the delivery tube, h, and heating this with a small flame. Steady ebullition is brought about by means of a fine piece of platinum wire, P, 0.04 mm. diameter, near the bottom of the still, connected to two thicker platinum leads,
1 Loc. cit.
F which pass through the side tube opposite the delivery tube and are connected with a battery of three or four accumulators. The fine wire is heated by the current of about 0.4 ampere, and a steady stream of bubbles is thus produced. This method is also recommended by Bigelow.- The receiver, b, is of the same form as Lehfeldt's, except that the tube below, instead of being provided with a stopcock, is bent, as shown in the figure, and is connected with a second small receiver, c, into which, by diminishing the pressure in the reservoir, f, the first small portion of distillate, before the temperature and pressure have become constant, is carried over. The distillate required for examination (about 1 c.c.) is then collected in b and, after admission of air, c is removed, and, by slightly raising the pressure, the distillate is forced out of B into a little test tube.
The arrangement of the manometer, the pump and the two air reservoirs, d and F, is shown in Fig. 26. By means of the various stopcocks, the pressure in F can be lowered a little below that in D, and air can be admitted into the apparatus, either through the stopcock 3 into d, or through the calcium chloride tube into c or b. A series of determinations was carried out in the following manner:-About 100 to 120 c.c. of one of the two substances was placed in a, and the pressure under which it boiled at the required temperature, t, was ascertained. A small quantity of the second liquid was then introduced into A, and the first distillation at the same temperature, t, carried out, the pressure being again noted. At the end of the distillation about 1 c.c. of the residual liquid in a was removed and placed in a small test tube for subsequent examination. A further small quantity of the second liquid was then added, and a second distillation was carried out as before, and these operations were repeated until the mixture in A became rich in the second substance.

Fig. 26. - Zawidski's apparatus.
1 Bigelow, "A Simplification of Beckmann's Boiling Point Apparatus," Amer. Chem.. Journ., 1899, 280.
The series of operations was then repeated, starting with the second liquid in a and adding small quantities of the first.
Rosanoff, Bacon and White's Apparatus. - The method employed by Rosanoff, Bacon and White1 was devised for rapidity of working and also to get over the difficulty that the composition of the first small fraction is liable to be seriously affected by the presence of even a trace of moisture in the liquid distilled.
The apparatus is shown in Fig. 27. It consists of a pear-shaped vessel with a long neck near the upper end of which are four circular openings for the escape of the vapour. A glass jacket, fused on to the rim of the neck, surrounds the flask and ends below in a tube through which the vapour escapes into a powerful worm condenser, and thence, in liquid form, into a receiver having several compartments for the convenient collection of consecutive fractions. The receiver communicates with the atmosphere through a tube filled with calcium chloride to keep out moisture. The neck of the pear-shaped boiling vessel is permanently stoppered above with a cork, which is made thoroughly vapour- and liquid-tight with shellac and sealing - wax. The cork carries an electric heater of platinum wire and, for the introduction and removal of liquid, an adapter tube reaching nearly to the bottom of the boiling vessel. Liquid is introduced with the aid of a separatory funnel as shown. The jacketed distillation vessel is all but completely immersed in a bath heated somewhat above the highest temperature to be attained by the boiling liquid. Reflux condensation in the distillation vessel is thus entirely prevented. In the final determinations a vessel of 125 c.c. capacity was used, and the shape of the platinum heater was such that a residue of barely 25 c.c. was left at the end of the distillation. The composition of the original liquid, the fractions and the residue was ascertained from their refractive indices.
The principle of the method may be best explained by an example. The original mixture of carbon disulphide and carbon tetrachloride contained 36.77 molar per cent of Cs2, the residue after distillation 22.18 molar per cent.
1 Loc. cit,

Fig. 27.
Continue to:


