9. Miscellaneous Dyes. Part 2
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
9. Miscellaneous Dyes. Part 2
200 gms. Hydrocyancarbodiphenylimide.
35 gms. H2S.
460 gms.
20%
Nh3.
25 gms. S.
(d) a-Isatin~anilide.
The ring-formation giving isatin derivatives only occurs under certain very definite conditions; it is important that hot sulphuric acid be used.
200 Gms. of finely divided dry thioamide are added during a quarter of an hour to 800 gms. of 94 % sulphuric acid, at exactly 940; the mixture heats up fairly strongly and must be cooled. As soon as all has been added the mixture is heated for a further hour to 106-108o, after which time the evolution of So2 will have ceased. The solution is cooled down to 200 and is converted directly into the hydrochloride of a-isatin-anilide by pouring in a thin stream into a mixture of 1 litre water, 2 kilos, ice, and 500 gms. salt, stirring being continued without interruption. The hydrochloride of isatin-anilide separates out, mixed with finely divided sulphur, as a light reddish-brown precipitate.
To obtain the anilide in a pure form it is filtered off and thoroughly washed with 20 % salt solution. The hydrochloride freed from all acid is then stirred up with water and dilute sodium carbonate solution
200 gms. Thioamide. 800 gms. 66° Be. H2So4.
1 litre H2O. 1 kg. Ice. 500 gms. NaCl.
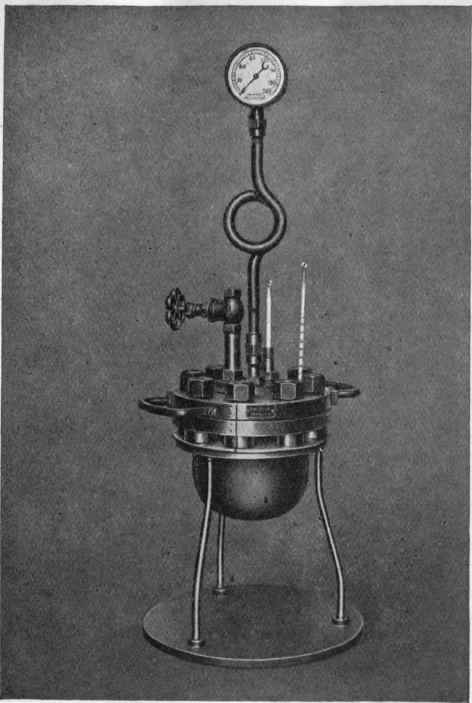
Fig. 35. - Cast-steel autoclave. Working pressure, 60 atms. Capacity, 1 litre. Weight, 30 kg.

Fig. 36. - Cast-iron autoclave with stirrer. Working pressure, 1 atm. Weight, 12 kg. Copper oil-bath.
Plate XIV.
until there is a faintly alkaline reaction. The precipitate of anilide and sulphur is filtered off, thoroughly washed, and the dried mixture extracted with cold carbon bisulphide. Finally, the anilide is crystallized from hot alcohol, from which it is obtained in the form of dark needles having a melting point of 126o, the yield from 200 gms. thioamide being about 150 gms. pure substance. On boiling with a slight excess of dilute hydrochloric acid, the anilido group is split off as aniline, pure isatin being precipitated directly. M.p. 200-201o. It is recrystallized from hot water in which it is easily soluble. Isatin is an important intermediate for the synthesis of various valuable vat-colours. Of particular importance are those vat-dyes obtained from a-isatin-anilide by condensing with β-hydroxy-thionaphthene (Thioindoxyl). G. Engi was the first to notice that quite different dyes are obtained according to whether isatin itself be used or its anilide. Isatin condenses by exchanging the β-oxygen atom whilst the anilide, curiously enough, splits off the aniline and gives a-condensation products. The a-condensation products are much more valuable than the isomers.
From Isatin:

Thioindigo Scarlet R
(Kalle). Ciba Red G = Dibrom derivative.
From a-Isatin-anilide:

Ciba Violet B = Tribrom derivative.
Ciba Violet 3B = Dibrom derivative.
Ciba Grey G =Monobrom derivative.
(e) a-Thioisatin and Indigo.
In order to obtain Indigo from the solution of isatin-anilide in sulphuric acid it is not necessary to isolate either the pure anilide or the hydrochloride, but the thioisatin may be obtained directly from the solution. A solution of sodium hydrosulphide is prepared by passing hydrogen sulphide into a solution of 45 gms. caustic soda dissolved in 150 c.cs. of water. This is then mixed with the sulphuric acid solution of isatin-a-anilide, from 200 gms. thioamide, by allowing both to run simultaneously into 6 litres of ice-water. A slight but distinct excess of sulphuretted hydrogen should always be present. The reduction occupies about half an hour, and the thioisatin separates out as a voluminous brown precipitate, aniline sulphate remaining behind in the solution. The thioisatin is filtered off as soon as a filtered test-portion of the liquid gives no further precipitate on treatment with sodium sulphide, which will be the case after about an hour. The precipitate is then washed until the mother-liquor has a specific gravity of only 1.007 (1° Be). The washed precipitate is stirred up with 3 litres of water to which concentrated sodium carbonate solution is added until the reaction becomes strongly alkaline, about 30 gms. soda being required for this purpose. The formation of Indigo takes place very rapidly, and the mixture is preferably warmed for one hour at 6o°, and is then left stirring over-night. Next day the Indigo and sulphur are filtered off, thoroughly washed, and dried at 8o°. The dried colouring matter is then extracted with twice its weight of carbon disulphide, 80 gms. pure Indigo being obtained.
Notes on Works Technique and Practice. - The reactions involved in the Sandmeyer synthesis go surprisingly smoothly, the yield of dye calculated upon the aniline taken being about 80 % of that theoretically possible. The process was used for a short time by J. R. Geigy, the cost of production being 10.8 francs per kilogram of 100 % product. Indigo prepared by the Sandmeyer method vats better than any other artificial product, and was immediately welcomed by dyers. It was found to be possible to effect the entire manufacture without the use of a drop of alcohol, since all the substances involved react readily in aqueous solution if sufficiently finely divided. The chief difficulty is not the hydrocyanic acid, but the hydrogen sulphide, which is a dangerous industrial poison, as, after a short time, it can no longer be smelt. The lead sulphide may be split up by means of concentrated hydrochloric acid into lead chloride and hydrogen sulphide, the latter being returned to the process. Soon after the introduction of this promising method of manufacture it was displaced by the process of the Deutsche Gold-und-Silber-scheide Anstalt, as in this latter method the yields were also increased to about 85 % by the latest improvements so that effective competition was no longer possible. At the same time it is always conceivable that under favourable conditions the Sandmeyer process
Continue to:


