9. Miscellaneous Dyes
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
9. Miscellaneous Dyes
Indigo (Traugott Sandmeyer's Method).1
Although Sandmeyer's Indigo synthesis is no longer worked, it affords such a classical example of the co-operation between science and technology that an account of it cannot be excluded from this book. This synthesis is one of the most remarkable achievements in the whole domain of dye chemistry, and may be compared with Leblanc's process for the manufacture of soda. Like the latter, it has exerted a fertilizing influence on the whole subject, and one portion of the process is still utilized for the production of isatin and its derivatives; these relationships will be discussed later.
Before describing the individual operations, the chemical mechanism of the reactions must be examined.
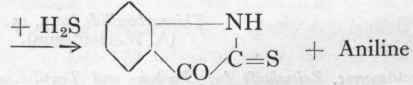
(a) Aniline is converted into A. W. Hoffmann's "thiocarbanilide" by heating with carbon bisulphide:

Thiocarbanilide (m.p. 151°) (A. W. Hoffmann).
1 See, also, Sandmeyer, Zeitschrift fur Farben- und Textil-Chemie, 1903, 7, 129; and Helvetica Chimica Acta, vol. ii., 234 (1919).
II
(b) The sulphur is removed from the Thiocarbanilide by means of basic lead carbonate, and at the same time hydrocyanic acid is added on, leading to the formation of Laubenheimer's "hydrocyancarbo-diphenylimide":

Hydrocyancarbodiphenylimide (Laubenheimer).
(c) The hydrocyancarbodiphenylimide is converted by means of yellow ammonium sulphide into the thio-oxamide-diphenyl-amidine, or more simply "thioamide":

M.p. 161-162o.
Thioamide.
(d) Under the influence of concentrated sulphuric acid the thioamide is converted readily into a-isatin-anilide:

a-Isatin-anilide. M.p. 126°.
(e) The a-isatin-anilide may be converted into Indigo in various ways; it is either reduced in alcoholic solution with dilute ammonium sulphide, or it is converted into the a-thio-isatin, which yields Indigo at once with alkalis. The last method has been chosen here as this was the process formerly made use of in the industry:
a-Thio-isatin,
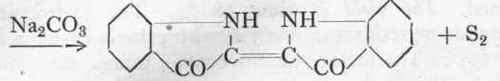
Indigo.
(a) Thiocarbanilide.
186 Gms. pure aniline are boiled up with 100 gms. of pure carbon bisulphide until the evolution of hydrogen sulphide ceases, which takes about 2 days. The temperature of the oil-bath is then raised to 160o, and the excess of bisulphide distilled off. The fused thiocarbanilide is poured on to a flat tray to cool, and is then powdered. It is sufficiently pure for further treatment, but may be obtained chemically pure by recrystallizing from three times its weight of alcohol. The yield is about 230 gms. M.p. 151°.
186 gms. Aniline. 100 gms.
Cs2
(b) Hydrocyancarbodiphenylimide.
350 Gms. lead nitrate, dissolved in a litre of hot water, are carefully precipitated at 95° with about 120 gms. dehydrated sodium carbonate and the precipitate is thoroughly washed with water. The moist basic lead carbonate is mixed with 600 gms. of 90 % alcohol in a 2-litre bolt-head provided with a stirrer and reflux condenser (Fig. 9), and is stirred up to a completely homogeneous paste; to it is then quickly added 228 gms. (1 mol.) of very finely powdered thiocarbanilide, and at 25° about 60 gms. of commercial sodium cyanide (=1.3 mol.).1 The temperature of the mixture must be raised during an hour to 700 with vigorous stirring, and a small test-portion is then filtered off. The colourless solution should no longer blacken a pinch of basic lead carbonate. If this is not the case, heating must be continued for a further hour and the test again applied; if complete desulphurization has still not been effected a little more lead carbonate and sodium cyanide may be added, but if the correct amounts of the reagents have been taken no further addition will be necessary.
As soon as the sulphur reaction is no longer given the mixture is heated up to boiling, and the hot solution is filtered off. The residue is extracted twice more with half a litre of alcohol, and the hydrocyancarbodiphenylimide allowed to crystallize out. The first fraction is quite pure, and weighs about 160 gms. After evaporating down the mother-liquor, a further 40 gms. of practically pure product
350 gms.
Pb(No3)2.
About
120 gms.
Na2Co3.
600 gms.
Alcohol.
228 gms. Thiocarbanilide. 60 gms.
NaCN.
1 The Hcn content of the sodium cyanide must be determined.
is obtained. The yield is about 98 %. The hydrocyancarbo-diphenylimide crystallizes in fine yellowish prisms, having a melting point of 137o. The mother-liquors contain prussic acid, and must be handled with due care.
(c) "Thioamide"
The addition of hydrogen sulphide to the hydrocyancarbo-diphenylimide takes place readily if it is very finely powdered, for which reason it is necessary to convert the product into the desired form either by grinding or by sifting. 200 Gms. hydrocyancarbo-diphenylimide are emulsified with 500 gms. yellow ammonium sulphide solution at 35o by vigorous stirring. The ammonium sulphide solution is prepared by passing 35 gms. hydrogen sulphide into a suspension of 25 gms. powdered sulphur in 460 gms. of 20 % ammonia. If the hydrocyancarbodiphenylimide has been powdered sufficiently, the hydrogen sulphide adds on quantitatively within 12 hours, which may be recognized by the fact that a washed test-portion of the product dissolves in dilute hydrochloric acid. It is then filtered off and thoroughly washed with water; the product is sufficiently pure for the next stage. Yield about 220 gms. It crystallizes from alcohol in yellow prisms having a melting point of 162o.
Continue to:


