Anthraquinone From Anthracene
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
Anthraquinone From Anthracene
Reaction:
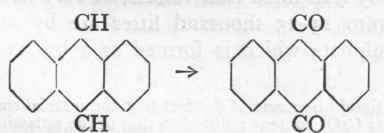
Anthracene.
Anthraquinone.
The anthracene used for the preparation of anthraquinone should not be too impure, or too much chromic acid will be used up. At the present day, the tar distilleries deliver a product of 80-92 % purity, which is estimated by the recognized methods (cf. Lunge, "Untersuchungsmethoden"). The commercial product is crystallized from pyridine.
Before oxidizing, the anthracene must always be sublimed by means of superheated steam at about 2000, as only in this way can it be reduced to a sufficiently fine state of division for use.
300 Gms. moist sublimed anthracene, calculated as 100 % product, are stirred up with 6 litres of water in a large lead-lined iron vessel and 600 gms. sodium bichromate are dissolved in it at the same time. The mixture is heated to 8o° by means of a Fletcher burner, and 1800 gms. 50 % sulphuric acid are run in from a dropping funnel during 10 hours. The presence of chromic acid must always be clearly shown, and the mixture must be stirred by means of a glass or wooden stirrer; finally, the mixture is boiled up for 2 hours, replacing the evaporated water. The product is filtered off and thoroughly washed. The mother-liquor may be worked up for chrome alum or for chromic sulphate.1
The thoroughly dried crude anthraquinone still contains some unchanged anthracene together with other impurities, and is carefully purified before working up further. Most of the impurities are removed by partial sulphonation, the pure product being finally redistilled with superheated steam.
The powdered and dried crude anthraquinone is heated with two and a half times its weight of 6o° Be. sulphuric acid to 120o, so long as sulphurous acid is evolved. After about 3 hours the mixture is poured into about three times its weight of water, filtered and thoroughly washed. The purified anthraquinone is then sublimed with steam at 240-260o. It is obtained as a fine faintly yellow powder (for apparatus see Fig. 17). The yield of dried product obtained from 100 gms. pure anthracene is about 106 parts of sublimed 100 % anthraquinone.
Notes on Works Technique and Practice. - The oxidation of anthracene is carried out in the works in lead-lined wooden vessels, or homogeneously lead-lined iron vessels, of very large dimensions. Vats holding from 15-25 thousand litres are by no means rare. The chromic sulphate which is formed as a by-product plays an
300 gms. 100 % Anthracene.
600 gms. Na2Cr2O7. 6 1. H2O.
1800 gms.
50%
H2So4.
1 Careful note should be made of the fact that commercial sodium bichromate nearly always has its CrOa content reduced to that of the potassium salt by means of Glauber salt.
important part in calculating the cost of the product, as it finds use for the chrome-tanning of leather.
Attempts to obtain anthraquinone by other means, such as nitrous oxides and air, have failed, not on account of any special technical difficulties, but owing to purely business considerations. The B.A.S.F., for instance, attempted the oxidation of anthracene with N2O3 in the form of vapour, but had to go back to the old process after a short time, as their leather customers had to be provided with chromic sulphate without fail, and it was not possible to obtain it so cheaply by any other method. The Fabrik Griesheim Elektron are said to carry out the new method with success. This process might also, under some conditions, be of importance, as it is independent of the use of foreign chrome-iron ore. Should the chrome leather tanning be displaced by the newer synthetic tanning materials, then it is quite certain that the chromic acid method would in time gradually disappear.
The distillation of anthracene and anthraquinone is carried out in apparatus very similar to that required for diphenylamine (cf. p. 99). The vapours, however, are condensed in large chambers, about 3X3X5 metres, by spraying in cold water. The bottom of the chamber is covered with fine calico, which allows the water to run off, but retains the sublimate.
Continue to:


