Sulphanilic Acid. Bake Process
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
Sulphanilic Acid. Bake Process
So far we have only examined cases of sulphonation where the liquid was kept in motion by means of a stirrer. There is, however, another method of sulphonating, which is based on a quite different idea; certain substances sulphonate when their acid sulphates are heated to moderate temperatures. This method obviously applies only to bases, such as aniline, whilst benzidine and more complicated bases, such as dehydro-thiotoluidine, give by this method different isomers from those obtained with the liquid acid.
This reaction is usually referred to as the Bake Process, as the acid sulphates are heated on tin trays like baking tins to moderately elevated temperatures.
It is sufficient to heat the dry acid sulphates in thin layers at 170-210° for 5-10 hours in order to obtain practically quantitative yields of the desired sulphonic acids. The most favourable temperature must, of course, be first determined in each case. Again, certain bases, such as benzidine, toluidine, etc., carbonize very easily if any excess of sulphuric acid be taken, particularly in the presence of air; further, sulphones and disulphonic acids may be formed as well. In modern factories, therefore, the heating is carried out in vacuo, the sulphonation then going more smoothly and more quickly. The ovens used for this reaction are either directly heated with fire, or, better, with superheated steam. Electrical heating may also be employed, and has the advantage of easy regulation, besides avoiding the necessity for using thick boiler-plates.
Reaction:
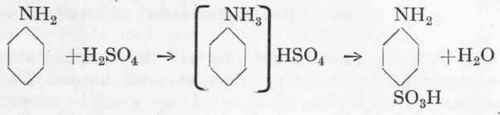
Sulphanilic Acid. - 105 gms. (=1 gm.-molecule) of 66° Be. sulphuric acid is mixed with 93 gms. aniline (=1 gm.-molecule) in an iron basin, the base being placed in the vessel and the sulphuric acid added in a thin stream with good stirring. In the factory it is done in an iron pot, and may be worked over with an iron rake. The resultant thick paste is at once spread, whilst still hot, on iron trays (15x15 cms.), furnished with rims 2 cms. deep. The layer should be about 1 cm. thick (8 cms. on the large scale), and the trays are then placed in the drying chest at least 5 cms. from the heating surface, the latter being heated by means of a Bunsen burner fitted with a "mushroom" top; the mass is heated for 8 hours at 1900. The cakes are then removed from the oven and the resultant sulphanilic acid shaken out of the tin. It is about 90 % pure and pale grey in colour; in addition to sulphanilic acid it contains about 3 % of unchanged aniline and a little free carbon. For many purposes this crude sulphanilic acid may be used directly by dissolving in sufficient soda to give a strong blue coloration with litmus, in the present case some 60 gms. soda and 500 c.cs. water being required. The liquid is heated to boiling, water being added to balance evaporation, until the steam has removed the easily volatile aniline. It is then run through a cotton filter, and the solution contains a sulphanilic acid which will answer most of the technical requirements without further treatment. In order to obtain pure sulphanilic acid from the solution it is acidified with sulphuric acid until acid to Congo paper. The sulphanilic acid is precipitated in a very pure form which is, however, not adequate for analytical purposes (cf. Analytical portion).
105 gms.
H2So4,
66° Be. 93 gms. Aniline.
60 gms. Na2Co3. 500 gms. H2O.
ca. 55 gms. H2So4, 66° Be.
The yield of crude substance is approximately 175 gms., or about 140 gms. of purified reprecipitated acid.
Naphthionic acid (naphthylamine sulphonic acid 1:4) may also be prepared by the baking process in a similar manner. In this case, however, the sulphonation and further working up will not go so smoothly; from 5-10 % of the naphthylamine always remains unchanged but cannot, of course, be removed simply by distilling off with the water as in the case of sulphanilic acid. Further, the unchanged base cannot be removed by filtering the sodium naph-thionate solution as the base emulsifies in the solution of the salt and goes through all forms of filter. It is therefore necessary to dissolve the crude acid in alkali and to remove the naphthylamine by treating the alkaline solution with benzene in an extraction apparatus. A further drawback is that a certain quantity (3-7 %) of the 1:5 acid is always produced together with the 1:4 acid. This so-called Laurent's acid can only be removed by crystallizing out the naphthionate, for which reason the naphthionic acid is always dealt with as its sodium salt (naphthionate).
Continue to:


