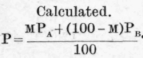Composition Of Liquid And Vapour Phases. Theoretical. Part 5
Description
This section is from the book "Distillation Principles And Processes", by Sydney Young. Also available from Amazon: Distillation Principles And Processes.
Composition Of Liquid And Vapour Phases. Theoretical. Part 5
Modification Of Brown's Formula
Brown's formula is not applicable to this pair of liquids, but by taking c =const. (c0)+aM instead of c = const., a good agreement between the observed and calculated values is obtained.
Mixtures of benzene with carbon tetrachloride have also been investigated by Zawidski with very similar results. He finds, however, somewhat lower vapour pressures for the pure substances, and his pressure differences are rather larger. The formula deduced from his data would be c = 0.961 +0.0036m, and there is a very fair agreement between the calculated and observed percentages of A in the vapour, though not quite so good as in the table on the previous page. The first formula for c would indicate the existence of a mixture of minimum boiling point containing 6.7 molecules per cent of benzene, the second a mixture containing 10.8 molecules per cent.
Distillations under Constant Pressure and at Constant Temperature. Of other pairs of liquids, the vapour pressures of which do not differ very greatly from those calculated from the formula there are two, carbon disulphide with carbon tetrachloride and carbon disulphide with benzene, which have been investigated by Brown,, the latter also by Carveth, but their distillations were carried out in the usual manner under constant pressure, not at constant temperature.
Brown observed, however, that the relation between m'A/m'B and mA/mB was the same for mixtures of carbon disulphide with carbon tetrachloride whether the distillation was carried out under a pressure of 432 mm., or under atmospheric pressure. If the values of c for other substances also are independent of, or vary only slightly with, the pressure, they can be calculated from the results of a distillation carried out either at constant temperature or under constant pressure.
It would not be safe to conclude, without further evidence, that the same values of c would be obtained in other cases by both methods, but we may at any rate assume that the differences would not be great enough to invalidate the general conclusions deduced from a comparison of the behaviour of different pairs of liquids.
In the cases so far considered, the composition of the vapour was determined experimentally. Such direct determinations have not yet been carried out for mixtures of chlorobenzene and bromobenzene, nor for those of any of the five pairs of closely related liquids mentioned on p. 32. Rosanoff, Bacon and Schulze,1 however, showed how the ratio of the partial pressures pA/pB and, in the case of substances of normal molecular weight, the molar percentages in the vapour could be calculated with accuracy from the vapour pressures of the pure components and the total pressures, P (p. 93).
They determined the vapour pressures of mixtures of benzene and toluene with great care at 79.7°, and their results, like those of Fortey and Young, show that these pressures are represented without serious error by the formula P = mPa + (1 - m)Pb, the maximum difference between the calculated and observed values of P being +1.9 mm. [Molar percentage of benzene in the liquid = 54.51 ; P (observed) = 537.5 mm.]
From their results and by means of their formulae, Rosanoff, Bacon and Schulze calculated the molar percentages of benzene, and these, which may be accepted with great confidence, are given in the table below.
1 "A Method of finding the Partial from the Total Vapour Pressures of Binary Mixtures, and a Theory of Fractional Distillation," J. Amer. Chem. Soc, 1914, 36, 1993.
Table 28. A=Toluene; B = Benzene; C = 2.589 ; t = 79.7° ; PB/PA at 79.7° = 2.5951
Molecular percentage of A in liquid. | Vapour pressures. | Calculated molecular percentage of A in vapour. | ||||
Observed. | | | R. B. & S. | Brown c = 2.589. | | |
100.0 | 748.7 | 748.7 | 00 | |||
95.65 | 729.0 | 728.7 | -0 3 | 98.27 | 98.27 | 00 |
91.89 | 711.4 | 711.4 | 00 | 96.72 | 96.70 | -0 02 |
82.43 | 668.0 | 667.9 | -0.1 | 92.49 | 92.40 | -0 09 |
7327 | 624.9 | 625.7 | + 0 8 | 87.82 | 87.65 | -0.17 |
63.44 | 579.2 | 580.4 | + 12 | 81.97 | 81.79 | -0.18 |
54.51 | 537.5 | 539.4 | + 1.9 | 75.74 | 75.62 | -0.12 |
43.52 | 487.0 | 488.8 | + 18 | 6656 | 66.61 | + 0 05 |
33.83 | 443.1 | 444.2 | + 11 | 56.76 | 56.97 | + 0 21 |
22.71 | 392.8 | 3930 | + 0 2 | 42.95 | 43.21 | + 0 26 |
11.61 | 341.5 | 341.9 | + 0 4 | 25.30 | 25.38 | + 0 08 |
0.0 | 288.5 | 288.5 | 00 | ... | ... | ... |
The chemical relationship between benzene and toluene is not quite so close as that between the members of the other five pairs of liquids, and the volume and temperature changes on admixture are noticeably greater, but, even so, it is found that Brown's formula is applicable without serious error to mixtures of these two substances and that the best value of the constant c differs but slightly from the ratio PB/PA
Continue to: