1. Sulphonations. Part 14
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
1. Sulphonations. Part 14
The pure sulphonic acids are distinguished by a very characteristic fluorescence, which can, however, only be seen clearly with very pure products, as otherwise they are hidden by the fluorescence of the 2:6:8 acid. The 2:6:S-naphthylamine disulphonic acid fluoresces blue, the 2:5:7 acid green, and the 2:1:5 acid red. Further, the 2:6:8 and 2:5:7 acids show a different behaviour with an acetic acid solution of diazotized nitraniline; the 2:6:8 acid gives only a faint yellowish coloration in dilute solution due to the formation of a diazoamino compound, whilst the 2:5:7 acid, on the contrary, yields at once a true red azo colour. It is therefore possible to gain an idea as to the purity of the products from the intensity of the colorations. Again, the diazotized 2:6:8 acid forms a very difficultly soluble red azo dye with R-salt which is precipitated at once even at great dilution and dissolves with a red colour on boiling, whilst the 2:5:7 acid gives an orange-red dye which is easily soluble.
Notes on Works Technique and Practice. - The addition of soda to the substance which is to be sulphonated is made solely for the purpose of preventing the formation of lumps on mixing with sulphuric acid; the mixture is broken up by the carbonic acid given off and very small quantities of soda suffice. In the factory the process is often carried out somewhat differently from that given above. Instead of sulphonating with monohydrate, the sulphate or the free B-naphthylamine base is added directly to 40 % oleum. Also less monohydrate is used as a diluent; the reasons for this are the same as those already given (cf. p. n). In a well-conducted works the isolation of the various sulphonic acids is comparatively easy, as the individual acids can be better separated when working with large quantities than in the laboratory. Filtration is usually carried out by means of wooden filter-presses fitted with so-called nitro-filters (q.v.). The purified acids, or their acid salts, may be centrifuged with advantage. The various mother-liquors, which on the laboratory scale contain an inseparable mixture of acids, are worked up either separately or mixed together according to the degree of purity. For this purpose they are neutralized completely with soda and "evaporated down to salt," that is to say, they are evaporated down in a multiple-effect vacuum concentrator until the sparingly soluble sodium chloride is precipitated out; the latter is then always centrifuged and the mother-liquors returned to the process. It is found that during the sulphonation a certain amount of diazotizable nitrogen always disappears ; this is to be attributed partly to the direct combustion of the substance, and partly to the formation of very easily soluble sulphones or sulphamides, the presence of which is readily noted owing to their yellow colour.
The 2:5:7 and 2:6:8 acids are melted with caustic soda to the corresponding aminonaphthol sulphonic acids, or, more rarely, they are sulphonated further. The 2:1:5 acid, however, is either worked up directly to light-resistant azo colours of the Lithol Red type, or it is sulphonated a stage further and is then fused to give aminonaphthol disulphonic acid 2:5:1:7.
The total yield of titratable naphthylamine disulphonic acids is very satisfactory. Approximately the following quantities are obtained from 1 molecule of β-naphthylamine or from the corresponding quantity of β-naphthylamine sulphate :
1 Molecule (=69 gms. sodium nitrite) yields about | ||||||
29 nitrite as 2:6:8-naphthylamine disulphonic acid | ||||||
20 | ,, | 2:5:7 | ,, | ,, | two separate fractions. | |
3.5 | ,, | 2:1:5 | ,, | ,, | ||
II | ,, as residual acids=mixture of various isomers. | |||||
Total=63.5 | nitrite as definite disulphonic acids=92 % of theory. Products equivalent to about 3.5 nitrite remain unaccounted for. | |||||
Aminonaphthol Sulphonic Acid 2:8:6 (y-Acid). M.W. 239.
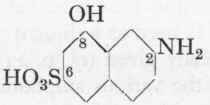
The melt of the pure naphthylamine disulphonic acid offers no special difficulties so long as the acid is as free as possible from sodium chloride (cf. p. 22).
A quantity of naphthylamine disulphonic acid 2:6:8 equivalent to 35 gms. nitrite (either the pure dry substance or the corresponding amount of moist acid) is heated with 220 gms. of chlorate-free caustic soda and 120 gms. of water in a stirring autoclave during 7 hours at 205-210°, the pressure rising to 14 atmospheres. After cooling and releasing the pressure the contents of the autoclave are diluted up to 1 litre (N.B. the melt should not smell strongly of ammonia), and concentrated sulphuric acid is added until distinctly mineral acid, about 250 gms. cone, sulphuric acid being required for this purpose. After standing a few hours the Gamma-acid is filtered off and well washed with cold water, in which it is very sparingly soluble; the cake is then pressed and dried at 100°. .
35 gms. Nitrite. 2:8:6-Naphthyl-amine disulphonic acid. cα. 180 gms. 220 gms. NaOH. 120 gms. H2O.
Yield: About 105 gms. (=95 gms. of 100 % product) Gamma-acid from "35 gms. nitrite" or approximately 80 % of theory. The acid is estimated by coupling with Normal diazotized aniline in dilute strongly alkaline solution, and simultaneously in another sample by diazotizing in very dilute mineral acid solution (for general details as to this type of estimation see Analytical Section). The figures obtained for the two estimations should agree within 1 %, as in the case of H-acid. If the melt has been carried out at too low a temperature the nitrite number will be greater than the coupling figure. The γ-acid should be at least 91 %.
250 gms. conc. H2So4
Aminonaphthol Sulphonic Acid 2:5:7 (J-Acid, iso-γ-Acid). M.W, 239.
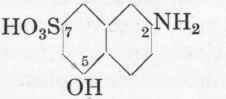
The method is exactly the same as for Gamma-acid, except that preferably somewhat more water is used for the melt, namely 160 gms. instead of 120, and further the temperature is a trifle lower, 200-205° for 7 hours.
The yield is about the same as in the case of γ-acid, i.e. about 95 gms. of 100 % aminonaphthol sulphonic acid 2:5:7 (=circa 105 gms. at 92 %) or 82 % of theory. The yield of 2:5:7-acid is therefore a trifle better than that for γ-acid.
Continue to:


